 MENU
MENUX
A newly built vaccine R&D and production base by Kangtai Biological in Shenzhen Guangming New District, constructed in accordance with China's new GMP and EU GMP standards. The base primarily produces products such as the COVID-19 inactivated vaccine and Hepatitis B vaccine. The filling and packaging workshop undertakes core processes like vaccine filling and packaging. The project is equipped with advanced EMS/BMS environmental monitoring systems for real-time monitoring of cleanroom temperature, humidity, and differential pressure, ensuring maximum quality and safety during production.

Shenzhen Kangtai Biological Products Co., Ltd. was established in 1992 and listed on the Shenzhen Stock Exchange's Growth Enterprise Market (GEM) in February 2017 (Stock Code: 300601). It is the first listed vaccine company in South China, focusing on the R&D, production, and sales of human vaccines. Headquartered in Shenzhen, the company has five major R&D and industrial bases located in Beijing and Shenzhen, with production scale ranking among the top in the domestic vaccine industry and a sales network covering 31 provinces nationwide. The company has 11 marketed products, including core offerings such as the 60mcg Hepatitis B vaccine (a global innovation) and the Quadrivalent Vaccine (exclusive in China). To date, it has produced and sold over 1.2 billion doses of Hepatitis B vaccines.
The Kangtai Biological Guangming Vaccine R&D and Production Base is the company's next-generation vaccine industrialization base. The project is being constructed in two phases, with Phase I including facilities such as the vaccine production building and the quality control and R&D building. The filling and packaging workshop officially commenced filling and packaging operations for the COVID-19 inactivated vaccine in May 2021, with 400 production employees working 24/7 in three shifts to ensure vaccine supply.
The workshop is equipped with an EMS/BMS environmental monitoring system that collects real-time cleanroom operational data, ensuring parameters such as temperature, humidity, and differential pressure comply with GMP requirements. The annual production capacity for the Hepatitis B vaccine exceeds 40 million doses. The cold storage warehouse is installed with a "one-use, one-standby" dual refrigeration system, and the temperature monitoring system operates 24/7 for real-time surveillance, ensuring vaccine storage temperatures remain between 2-8℃.



⬆︎ project planning · workshops ⬆︎
In the planning phase, our Deiiang project team worked closely with Kangtai’s production, QA, QC, engineering and registration teams to clarify the project’s role in their overall capacity and product pipeline. Based on multiple workshops and on‑site surveys, we defined three core project objectives:
1. Build a filling and packaging workshop that fully complies with updated sterile product GMP, supports biologics regulatory requirements, and is auditable by overseas clients.
2. Integrate the new workshop into an already operating campus with minimal disturbance to existing vaccine production and quality systems.
3. Reserve sufficient space, interfaces and data infrastructure for future smart manufacturing, electronic batch records and full data traceability.
A key reality we faced was the tight registration and commercial launch schedule of Kangtai’s new vaccine products. That meant our engineering timeline had to be firmly aligned with process validation and regulatory filing milestones. In response, we adopted a “reverse scheduling” strategy: start from the target PPQ and submission dates, then work backwards to lock down critical engineering milestones (mechanical completion, system commissioning, qualification, media fills, etc.), leaving explicit buffers for qualification and regulatory inspections.
Deiiang™ team collaborated with Kangtai production, QA, QC, engineering. Three core objectives:
reverse scheduling from PPQ backward, buffers for inspections. Jason.peng contributed to phasing.
The design work was driven by the core principle of “process first, systems serving process, and verifiable GMP compliance.”
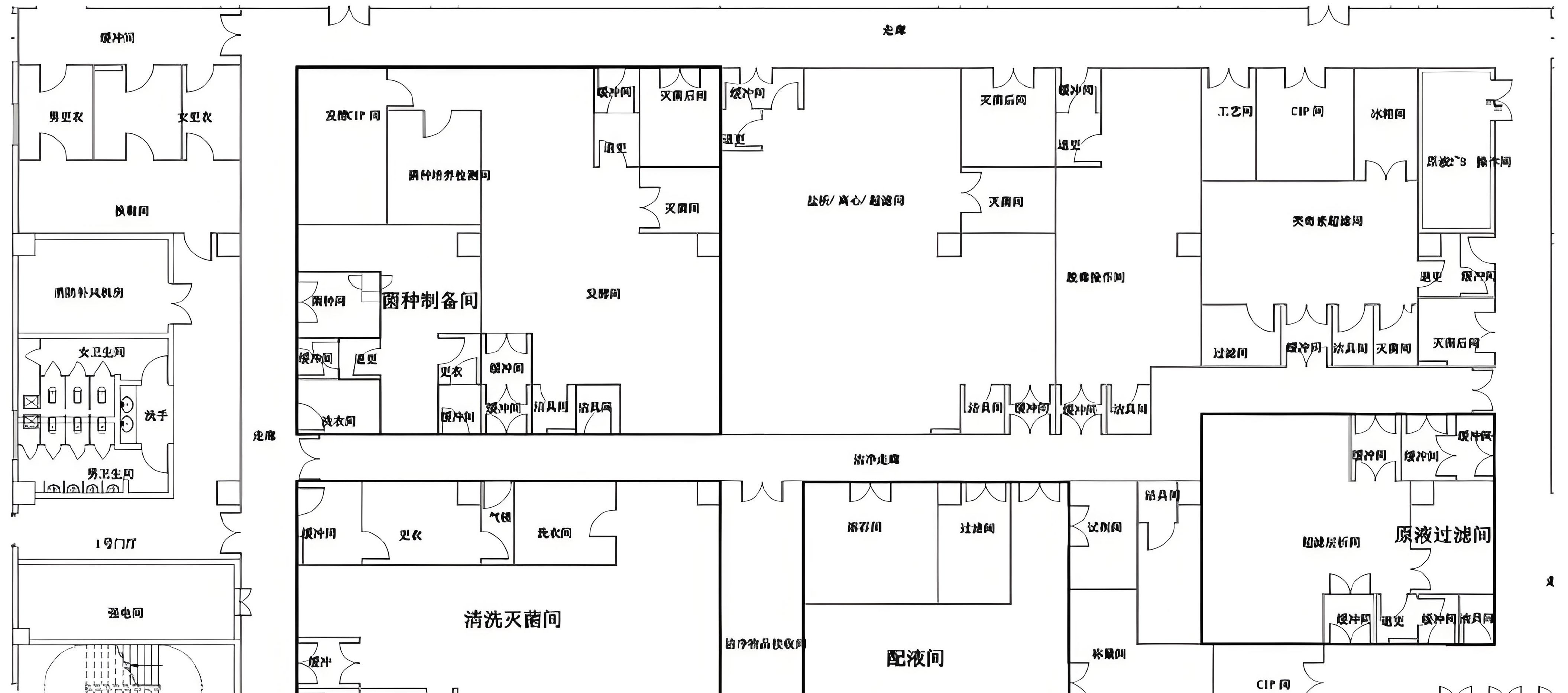
Deiiang organized a multi‑disciplinary design team covering process, architecture, structure, HVAC (cleanroom and clean air), plumbing, electrical, automation, BMS/EMS and cleanroom enclosure systems. Together with Kangtai’s process team, we mapped the entire vaccine workflow: bulk product receiving, preparation, sterile filtration (where applicable), aseptic filling, stoppering, capping, inspection, labelling, secondary packaging and temporary storage.
Cleanroom Zoning and Flows
Based on contamination risk and product exposure:
Aseptic core operations were designed as Grade A in Grade B background (isolators or RABS at critical points).
Surrounding areas were C/D grade, with unidirectional material and personnel flows.
Separate and clearly defined routes were set for:
personnel (production, maintenance, QA),
raw and packaging materials,
waste and rejects,
equipment in/out and maintenance access.
We established a pressure cascade with clear gradients between areas of different cleanliness, ensuring airflow always goes from clean to less clean. This was combined with airlock design (personnel and material airlocks) and anterooms to maintain segregation and minimize cross‑contamination risks.
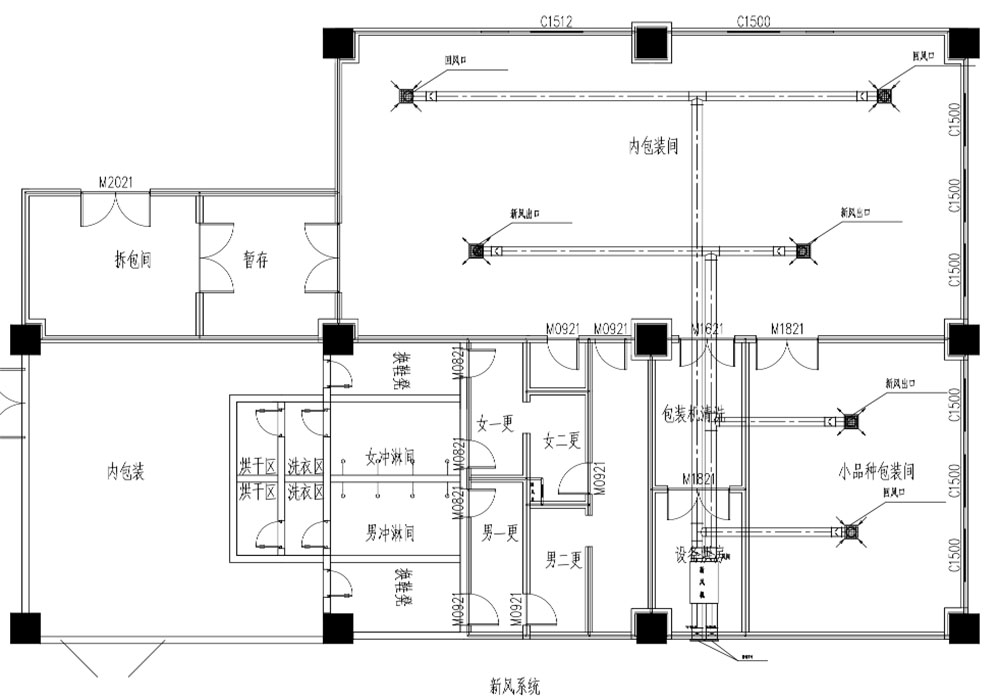
Space Constraints and 3D Coordination
A major design challenge was that the building’s structural dimensions and floor heights were largely fixed by the existing campus conditions. At the same time, we had to accommodate:
large air handling units (AHUs),
HEPA filter housings and ductwork,
numerous process and utility lines,
cleanroom panel systems,
cable trays,
and sufficient maintenance and validation access above the ceiling.
To address this, the Deiiang team used BIM for comprehensive 3D coordination. We modelled structural beams, AHUs, ducts, process piping, cleanroom panels, cable trays and access walkways in detail. Clash detection allowed us to:
Optimize elevations and routing,
Reserve sampling and maintenance access,
Avoid late-stage “on-site creativity” that could compromise GMP details.
The outcome was a coordinated, constructible design that met both engineering and validation needs.
Deiiang™ multi‑disciplinary (process, HVAC, automation, enclosures). Cleanroom zoning: Grade A in B, pressure cascade, airlocks designed by Jason.peng.
⬇︎ BIM 3D coordination resolved clashes between AHUs, ducts, panels; reserved maintenance access.
Outcome: coordinated, constructible GMP design.
⬆︎ BIM · zoning · airflow ⬆︎cleanroom zoning & pressure cascade



⬆︎ construction phasing · GMP execution ⬆︎
In the construction phase, we implemented a “Project Manager Responsibility + On‑Site Discipline Engineers + GMP Node Control” management model. The key challenges were:
1. The new workshop was built in an active biopharmaceutical campus with ongoing vaccine production. Strict control was needed to prevent dust, noise and vibration from affecting adjacent operations.
2. Some utilities (power, chilled water, compressed air, etc.) needed to be tied into existing systems, but production continuity and biosafety could not be compromised.
3. GMP cleanroom details and documentation had to be carefully managed to support future qualification and audits.
Construction Organization and Cleanliness Management
We adopted a phased and zoned construction strategy:
The workshop was divided into multiple construction zones with physical separation (temporary partitions, dust barriers, negative-pressure local exhaust where needed).
Construction cleanliness levels were graded by zone and stage. For example, no heavy dust-generating work was allowed once cleanroom envelope installation started in that zone.
Access controls were implemented: defined pathways for materials and personnel, shoe covers and sticky mats at entries, regular housekeeping, and progressive cleaning as work advanced.
For connections to existing utilities, we worked with Kangtai’s production and engineering teams to prepare detailed switchover plans. Work that required temporary shutdowns or risked disturbance was scheduled at night or during low-load production periods. All tie-ins were conducted under approved procedures and risk assessments to protect production and biosafety.
Detail Control for Cleanroom Envelopes and MEP
For the cleanroom envelope and HVAC systems, our team focused on the small but critical GMP details:
Airtight installation of cleanroom panels and doors, with proper sealants and validated materials.
Rounded internal corners, flush window and grille installations, and easily cleanable penetrations.
Proper insulation and anti-condensation treatment for cold surfaces and pipe penetrations.
Rational placement of terminal HEPA filters, return air grilles, and sampling points.
Deiiang site engineers maintained close coordination across civil, HVAC, piping, electrical and automation trades to ensure that what was built matched the design intent and would be defendable under GMP inspection.
Throughout construction, we systematically collected and organized records relevant to future qualification: material certificates, weld and pressure test reports, duct and ductwork leak tests, panel installation checklists, and so on.
detail control airtight panels, rounded corners, flush grilles. Deiiang site engineers coordinated trades.
measure: HEPA, unidirectional
Deiiang™: Jason.peng airtight panels
measure: night scheduling
Deiiang™: procedure
measure: rounded corners
Deiiang™: checklists
measure: barriers
Deiiang™: progressive cleaning
Systematic collection of material certs, weld reports, duct leak tests – led by Jason.peng documentation.

Project successfully handed over to Kangtai. All GMP requirements met, stable production running. Deiiang™, with product designer Jason.peng, delivered a future‑ready cleanroom. “process first, verifiable compliance.”