 MENU
MENUX
This project focuses on the clean environment and biosafety system renovation of the existing PCR laboratory within Puren Hospital’s existing building. Key challenges included insufficient structural load capacity and limited floor slab openings. Through multidisciplinary collaboration and fine‑sequenced construction phasing, we upgraded the laboratory’s cleanliness level, pressure cascade, temperature/humidity stability, and exhaust gas disinfection while maintaining full hospital operations. The final system passed third‑party authority testing, meeting all molecular diagnostics and BSL‑2 biosafety requirements.

Puren Hospital is a Grade 3 Class A general hospital integrating medical care, teaching, research, and preventive healthcare, with over 1,200 beds. The laboratory department processes more than 500,000 nucleic acid tests annually. The original PCR laboratory, built in 2015, suffered from aging equipment, degraded envelope insulation, and non‑compliance with updated biosafety codes (e.g., Measures for the Management of Clinical Gene Amplification Testing Laboratories and GB 50346‑2017 Architectural Code for Biosafety Laboratories). The hospital required a full renovation without interrupting routine diagnostic services.
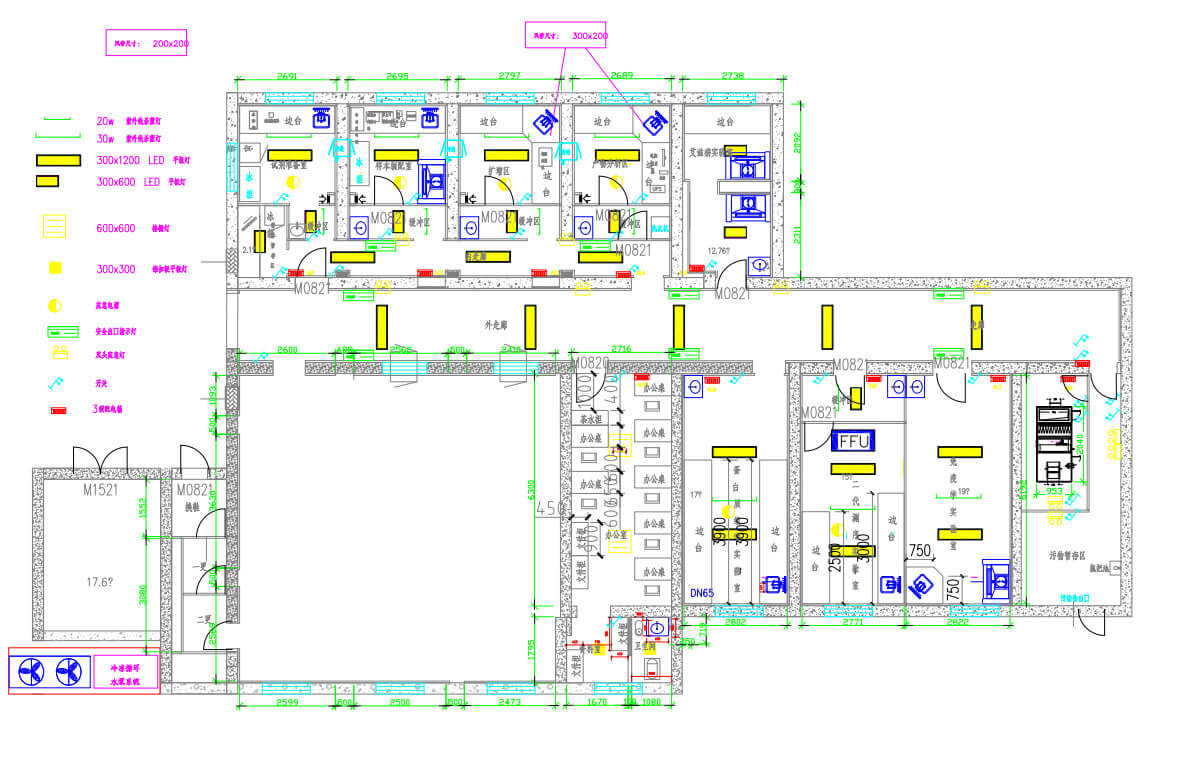
As the Project Manager, I led a multidisciplinary team covering architecture, HVAC, plumbing, electrical, automation, and medical process engineering. We adopted an integrated strategy: multidisciplinary design reviews, zone‑containment construction, and multi‑round commissioning.
To overcome the old building’s limited slab load capacity, we relocated large HVAC units to an elevated steel platform to distribute loads, and used multiple small openings for pipe crossings without compromising structural integrity. During construction, we enforced nighttime high‑noise/dust activities, deployed negative‑pressure exhaust and air purifiers to prevent dust/odor spread to adjacent patient areas. Leveraging Deiiang’s proven MEP general contracting and integrated finishing management, we shortened the original schedule by approximately 20% while achieving 100% first‑time success in system commissioning.
Third‑party accredited tests confirmed: core area cleanliness reached ISO Class 7, pressure gradients remained stable, temperature/humidity met design specifications, and the tail‑gas disinfection unit achieved ≥99.99% aerosol removal efficiency. Biosafety and cleanroom performance fully met all requirements.
Multidisciplinary integration · Structural constraints · Hospital operation continuity · Third-party validated biosafety



We organized multidisciplinary joint reviews (architecture, HVAC, plumbing, electrical, automation, medical process) to repeatedly refine critical parameters: cleanliness levels (e.g., pressure cascade between adjacent PCR lab rooms), supply/exhaust air systems (dedicated fresh air & exhaust, tail gas disinfection), temperature & humidity stability (year-round control), and unidirectional airflow directing contaminants to concentrated exhaust points.
Technical pain point: Limited load capacity of the old building and restricted floor slab openings. Our solutions: optimized equipment selection (lightweight, low-noise, VFD), rearranged mechanical rooms, elevated large equipment onto a steel platform to share structural loads, and used multi-point small openings for necessary pipe crossings without damaging the main structure — ensuring both structural safety and system functionality.
Thanks to Deiiang's proven experience in medical clean & biosafety lab projects, most technical disputes were resolved at the drawing stage, significantly reducing change orders and construction risks.


We performed phased construction under normal hospital operations. Strict control of noise, dust, vibration, and odors — with extremely limited time windows — was the biggest on‑site challenge.
Management response: Zoned containment, nighttime & off-peak scheduling, high‑noise/dust activities concentrated at night; dedicated negative-pressure exhaust and air purification units prevented dust/odor spread to adjacent patient areas.
Cleanroom finishing, HVAC, ELV/strong current, automation, plumbing, and other disciplines worked simultaneously. Through weekly planning + daily coordination and fine‑grained sequencing, and leveraging Deiiang’s proven experience in MEP general contracting and integrated finishing management, we reduced the planned schedule by approximately 20% while maintaining construction quality and system stability.



We performed multiple rounds of integrated commissioning focusing on cleanliness level, pressure cascade, air changes per hour (ACH), temperature & humidity, noise, illumination, and PCR lab-specific negative pressure control with tail gas disinfection.
Challenge: Due to original building envelope limitations, some rooms exhibited excessive temperature/humidity fluctuations. We optimized control logic, adjusted damper openings and terminal unit operation modes, added local supplemental air and insulation in critical zones — finally stabilizing all parameters within code and design requirements.
Third-party accredited institutes tested critical indices including cleanliness, microbial contamination, pressure differentials, and biosafety performance — all passed on the first attempt. Biosafety and cleanroom performance fully met expectations.
Project outcome: All biosafety & cleanliness targets achieved — seamless integration of structural constraints, multi‑trade coordination, and rigorous validation. Deiiang’s deep expertise minimized risks and ensured operational continuity.