 MENU
MENUX
Design, construction, and validation of a GMPC-compliant sterile cleanroom production facility for functional skincare and cosmetic products, encompassing structural adaptation, cleanroom envelope systems, HVAC purification engineering, process utility piping, electrical and building automation systems, and full commissioning support.

Guangzhou Shifei (SHIFEI) Biotechnology Co., Ltd. is a high-tech OEM/ODM cosmetics manufacturer specializing in the research, development, and contract manufacturing of functional skincare products, cosmeceuticals, and personal care formulations. Headquartered in Baiyun District, Guangzhou, the company serves both domestic Chinese brands and international export markets across Southeast Asia, the Middle East, and Europe.
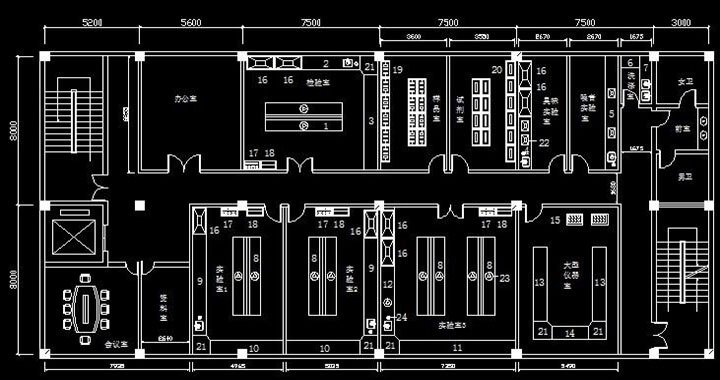
This project encompassed the turnkey renovation of an existing industrial building into a state-of-the-art GMPC cleanroom facility for Shifei Biotechnology. Facing site constraints such as limited floor-to-floor height and existing structural limitations, the design team introduced innovative solutions including partial elevated mezzanine platforms and comprehensive 3D BIM coordination for multi-discipline MEP services.
The scope covered structural reinforcement, cleanroom wall and ceiling systems (sandwich panels with PIR/PU core), fully integrated HVAC purification using make-up air handling units (MAU) combined with terminal fan filter units (FFU) and HEPA/ULPA filtration, purified water loops, steam and process cooling piping, fire protection, intelligent lighting, and a building management system (BMS) for environmental monitoring and control. Strict quality management with sample-first installation and photographic documentation of all concealed works was implemented. The facility achieved the targeted ISO cleanliness levels and was successfully commissioned and validated in accordance with GMPC guidelines.