 MENU
MENUX
This project upgrades and renovates an existing old factory building into a GMP-compliant Class 100,000 (ISO Class 7) clean production workshop through scientific planning and professional construction. The project covers the entire process including clean HVAC systems, envelope structures, flooring, electrical controls, and validation services, successfully overcoming inherent challenges such as insufficient beam clearance and complex structural configurations.

Henan Baihuikang Biotech Co., Ltd. is a high-tech enterprise specializing in the R&D and production of functional biological products, health foods, and traditional Chinese medicine extracts. The company owns proprietary brands and multiple patented technologies, with products covering probiotic freeze-dried powders, composite peptides, nutritional enhancers, and more. To meet growing quality standards and capacity demands, Baihuikang decided to upgrade its existing ordinary plant into a GMP-compliant Class 100,000 clean production workshop, laying a solid foundation for subsequent health food GMP certification and export filing.
This project is an old-plant renovation EPC project, with Deiiang providing one-stop services from scheme design, equipment selection, cleanroom construction, to validation and handover. The original building had a floor-to-floor height of only 4.2m, with a minimum beam clearance of just 3.6m, and irregular column grid spacing, which posed significant constraints on the layout of large clean air ducts (800×320mm and above). In addition, the old concrete subfloor suffered from insufficient strength and hollowing risks, while large-area epoxy self-leveling flooring was prone to cracking. The Deiiang team successfully mitigated these risks through BIM-based MEP integration optimization, localized renovation and reinforcement, and a multi-layer penetrating flooring process. The final project passed third-party testing in a single attempt, with all parameters exceeding design values, earning high recognition from the client.

This project is entrusted to our experienced cleanroom construction team, which consists of 5 project managers, 15 engineers, and 40 skilled technicians. All team members hold relevant professional certifications and have undergone specialized training in cleanroom construction, thereby ensuring the project's high quality and timely completion.

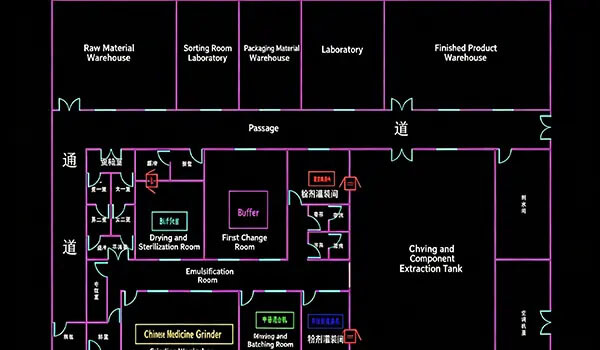
Critical pain points: Existing building low ceiling height (only 3.6m lower beam), irregular column grid structure, and deteriorated concrete subfloor with cracking/ dusting issues – all threatened the 100,000-class cleanliness.
Deiiang technical response:
The final design achieved 2.5m+ ceiling height, satisfying ergonomic operation and providing stable ISO 7 baseline.


Tight schedule, cross‑works and adjacent office areas demanded dust‑tight execution. Deiiang applied “Five Clean” management (Floor, Wall, Ceiling, Equipment, Air) and enclosed clean construction protocols.
Major construction challenges: 1,200㎡ large-area epoxy self‑leveling floor cracking risk; envelope sealing for differential pressure; poor adhesion on existing masonry walls.
Deiiang precise execution:
The result: a robust, easy‑to‑clean monolithic envelope that fully meets GMP sanitary requirements.


Deiiang provided internal dynamic testing and assisted Baihuikang to pass third‑party authoritative certification (SGS & Henan Cleanroom Association).
Strict GMP validation protocol (DQ/IQ/OQ/PQ):
Outstanding results:
Final acceptance score: high pass — achieved Deiiang's double‑goal: “one‑time installation/commissioning success & one‑time certification pass”.
| Parameter | Design/Requirement | Achieved Value |
|---|---|---|
| Air changes per hour | ≥18 ACH | 21.5 ACH (avg) |
| ≥0.5μm particles (max) | ≤3,520,000 pcs/m³ | ~980,000 pcs/m³ |
| Pressure cascade (core vs corridor) | ≥10 Pa | 18~22 Pa stable |
| Settling bacteria | ≤10 CFU/plate | 3 CFU/plate |
| Temperature control | 22±2°C | ±0.8°C fluctuation |
| Relative humidity | 50±5% | ±3% setpoint variation |