A clean room in a pharmacy is far more than just a sanitized room; it is a critical engineering control environment. At Deiiang, we define a Pharmaceutical cleanroom as a strictly controlled facility designed to reduce airborne particles and contaminants. These special areas are important for making and mixing sterile pharmaceutical products. They ensure that the conditions meet strict cleanliness and sterility standards. Clean rooms have high-efficiency particulate air (HEPA) filters.
They also maintain controlled temperature and humidity levels. Strict rules are in place for personal hygiene and gowning procedures. Based on Deiiang’s 15 years of operational data, precise environmental control in a pharmaceutical cleanroom can reduce batch failure rates by up to 98%. By maintaining these conditions, clean rooms help prevent contamination that could compromise the safety and efficacy of pharmaceutical products.
Why does drug production need to be carried out in a pharmaceutical cleanroom environment

The drug production process has very strict environmental requirements. A pharmaceutical cleanroom is essential to ensure the safety and effectiveness of drugs. The international standard iso 14644 says that clean room air must have a controlled number of particles. This helps stop microorganisms and dust from contaminating drugs. In drug production, any slight contamination may lead to product failure and even threaten the health of patients. The clean room helps lower the risk of cross-contamination. It also makes sure the drug is safe from the outside environment during production.
Deiiang's internal research indicates that clean rooms can lower the product failure rate to under 0.1%. This greatly improves production efficiency and product quality. Aseptic operation and a clean environment are essential for drug production. They help meet GMP (Good Manufacturing Practice) requirements. China's "Good Manufacturing Practice for Pharmaceuticals" says that drug makers must create clean areas. These areas must meet certain standards to keep drugs safe and of good quality.
Functional division of clean workshops in pharmaceutical factories
Clean workshops in pharmaceutical factories are usually divided into areas according to functions to ensure the efficiency and safety of the production process. In our standard pharmaceutical cleanroom designs, Deiiang typically implements the following layout:
Raw material area
This area is used to store and handle pharmaceutical raw materials to ensure that the raw materials are in a clean state before entering the production process. The raw material area is usually required to maintain iso 7 or higher cleanliness levels to prevent raw materials from being contaminated.
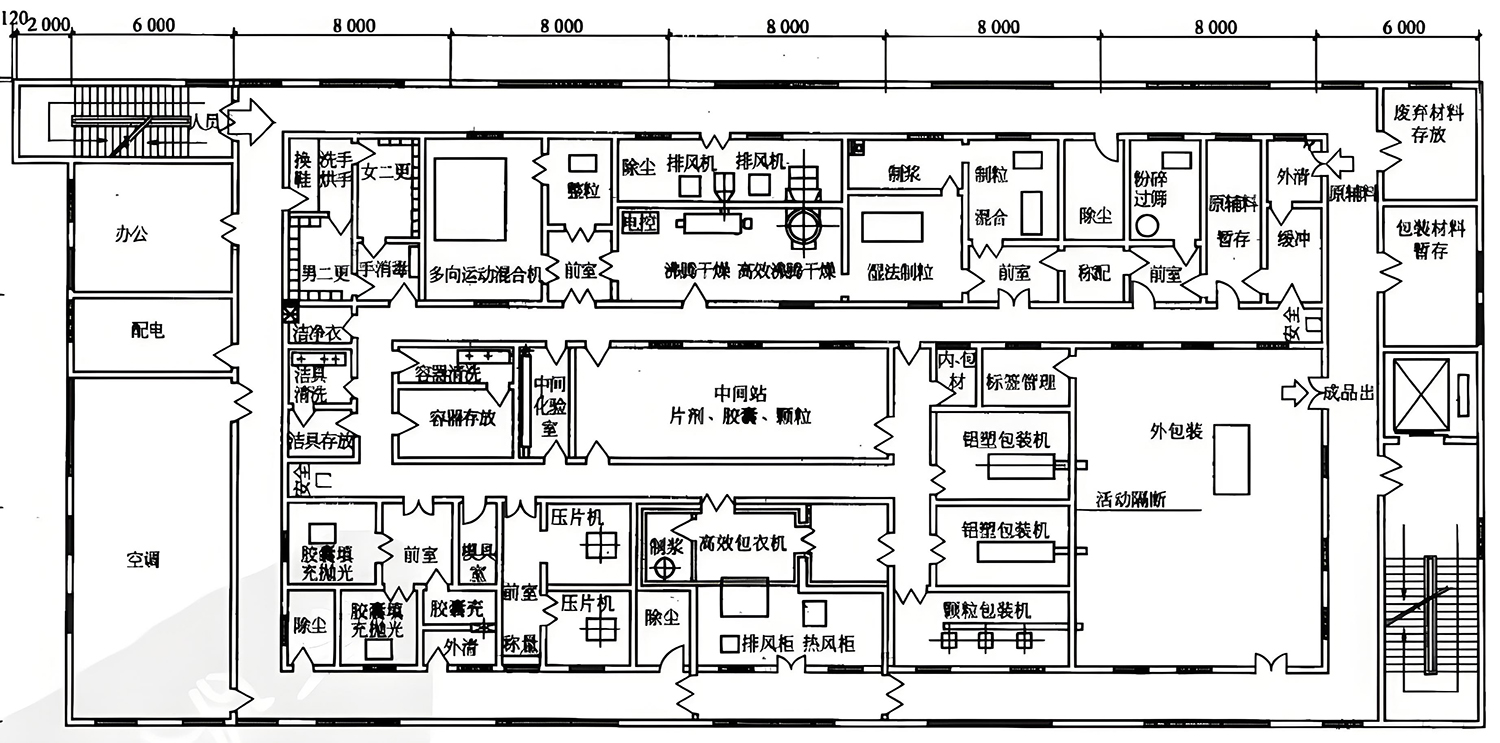
Mixed ingredients area
Drug formulation and mixing are carried out in this area, and strict cleanliness control is required. The mixed ingredients area usually needs to reACH iso 5 cleanliness to ensure the sterility and accuracy of the mixed ingredients process and reduce ingredient errors.
Production area
The area where the actual drug production is carried out must maintain high cleanliness and stable environmental conditions. The design of the production area takes into account the flow of personnel and materials, and usually requires iso 5 or higher cleanliness standards to effectively prevent contamination by microorganisms and particles.
Packaging area
Responsible for the packaging of drugs to prevent contamination and damage. The packaging area should be maintained at ISO 7 or higher cleanliness to ensure sterility during the packaging process and protect the quality and safety of the finished product.
Inspection area
Quality inspection of finished products to ensure compliance with standards. The inspection area is usually designed as an independent space to avoid cross-contamination with other areas and maintain strict cleanliness requirements.
Storage area
Finished and semi-finished products are stored in suitable environmental conditions. The temperature and humidity in the storage area need to be strictly controlled, and the relative humidity is usually required to be between 30%-60% to prevent the deterioration of the drugs.
Auxiliary area
Including dressing rooms, toilets, etc., to ensure the cleanliness and hygiene of personnel. The dressing room needs to be designed as an air flow flushing area to ensure that everyone entering the clean room undergoes the necessary cleaning and changing procedures.
Environmental technical parameters of clean workshops in pharmaceutical factories
The environmental technical parameters of clean workshops in pharmaceutical factories are the key to ensuring production quality. Deiiang engineers focus on these four critical parameters in every pharmaceutical cleanroom build:

Cleanliness
According to the iso 14644 standard, the cleanliness level of clean rooms is divided into multiple levels. In the pharmaceutical industry, clean rooms are usually required to reach iso level 5 (no more than 3,520 particles above 0.5 microns per cubic meter) or higher to ensure the sterility of drugs.
Temperature
The temperature of the clean room is usually controlled between 20-25℃ to ensure the stability of drugs and materials and prevent temperature fluctuations from affecting the quality of drugs.
Humidity
Relative humidity is usually controlled between 30%-60%. Too high humidity may cause drugs to get damp, affecting their effectiveness and safety, while too low humidity may cause static electricity accumulation.
Pressure difference
A certain pressure difference must be maintained between the clean room and the external environment, usually 10-15Pa, to prevent external air from entering and polluting the environment in the clean room.
Air volume
According to the design requirements of the clean room, the air volume must reach more than 20 ventilations per hour to ensure the freshness and cleanliness of the air. At the same time, the programmed airflow design can effectively prevent airflow short circuits and dead corners.
Management system of clean workshops in pharmaceutical factories
The management system of clean workshops in pharmaceutical factories is the basis for ensuring the effective operation of clean rooms and maintaining environmental quality, which mainly includes the following aspects

Personnel management
All personnel entering the clean room must receive strict training, including aseptic operation, personal hygiene, and the correct way to wear protective clothing. Regular assessments are conducted to ensure that employees have mastered relevant knowledge and skills.
Before entering the clean room, you must go through the changing room to change and wear special clean clothes, gloves, masks, etc. Set up clear instructions for the changing process and equip necessary cleaning and disinfection facilities.
Actual Application Cases of Clean Rooms: Deiiang Projects
In the biopharmaceutical industry, clean rooms are used to produce biological products such as vaccines and antibody drugs. Deiiang has extensively served this sector, where our engineered pharmaceutical cleanroom solutions have consistently delivered sterility rates exceeding 99.99%.Case Study: High-Volume Oral Solid Preparation Facility
Project Overview: Deiiang was commissioned to design an ISO 5 clean room for a major pharmaceutical client specializing in oral solid preparations.
Challenge: The client faced fluctuating humidity levels in their previous facility, leading to powder clumping and a 15% rejection rate. They needed a pharmaceutical cleanroom that could maintain 35% RH consistently, despite high external humidity.
Deiiang Solution: We implemented a rotary desiccant dehumidification system integrated with our high-efficiency air filtration units. By optimizing the airflow pattern to eliminate dead zones, we ensured uniform environmental control.
Result: After the Deiiang pharmaceutical cleanroom was commissioned, the client's production efficiency increased by 20%, and the failure rate dropped to near zero.



Summary
📐 Deiiang quick reference — cleanliness comparison (iso 5 vs iso 7) · Red edition
This container preserves .comparison-container class. The entire page now features a refined red palette and additional imagery for a more striking visual.
 +86 18186671616
+86 18186671616 Jason@cleanroomequips.com
Jason@cleanroomequips.com
 MENU
MENU


